
This discovery was very useful to modify the design of the cathode tubes used to date, and place concave cathodes in their corners, to produce focused rays that would be used in a variety of applications in the future. Through experiments on evacuated tubes, Goldstein detected that cathode rays could project acute shadows of emission perpendicular to the area covered by the cathode. Likewise, the essays of Eugen Godlstein also contributed in a remarkable way to deepening the technical notions about cathode rays. Hence, that this type of phenomenon is known in the scientific world as channel rays, since they pass through the existing perforation in the cathode of the study tube. The positively charged ions that make up the channel rays move towards the perforated cathode until they pass through it, given the nature of the experiment. Goldstein called these rays"Kanalstrahlen", to refer to the counterpart of cathode rays. These cations are attracted to the negative electrode of the tube, given the natural attraction between opposing electric charges. Gas atoms lose their negative charge, and are positively charged. Starting from this base, the electrons of the cathodic ray repel the electrons of the atoms of the gas, and these last ones are detached from their original formation. The channel raysĬhannel rays arise when the cathode rays collide with the atoms of the gas that is confined inside the test tube. That is, the channel rays had a positive charge. Because the channel rays traveled in the opposite direction to the cathode rays, Goldstein inferred that the nature of their electrical charge must also be the opposite. This new flow was baptized by Goldstein as channel rays. In addition, the behavior of the particles with respect to their electric field and magnetic field, was totally opposite to that of the cathode rays. Goldstein concluded that, in addition to the cathode rays, which traveled from the cathode (negative charge) to the anode (positive charge), there was another ray traveling in the opposite direction, that is, from the anode to the cathode of the modified tube. However, the highlight is that these rays moved in the opposite direction to the cathode rays and were called channel rays. Under this new configuration, Goldstein discovered that the tube emitted a new glow that started from the end of the tube that had been perforated. In addition, he repeated the experiment with the modification of the Crookes tube, increasing the tension between the ends of the tube to several thousand volts. Goldstein modified the structure of the Crookes tube, and added several perforations to one of the tube's metal cathodes. With this experiment the existence of elementary particles with negative charge in the atoms was demonstrated. Consequently, the gases become fluorescent when the circuit between the two ends of the tube is closed.Ĭrookes concluded that this phenomenon was due to the existence of cathode rays, that is, flow of electrons. When electrifying the tube, the air ionizes and becomes a conductor of electricity. The apparatus has two metal parts, one at each end, which act as electrodes, and both ends are connected to external voltage sources. The pressure of the gases inside the tube is regulated by moderating the evacuation of the air inside it. The base structure of the Crookes tube consists of an empty tube made of glass, inside which gases circulate. He then made modifications to the structure developed by William Crookes in the 19th century. Goldstein began his experiments with Crookes tubes during the decade of the 70s. 
Experiments with cathode rays Crookes tubes Nevertheless, Ernest Rutherford is considered the discoverer in the scientific community. On the other hand, he is sometimes credited as the discoverer of the proton, which I observe in the vacuum tubes where he observed the cathode rays. Goldstein did not propose an atomic model, although his discoveries allowed the development of the atomic model of Thomson. 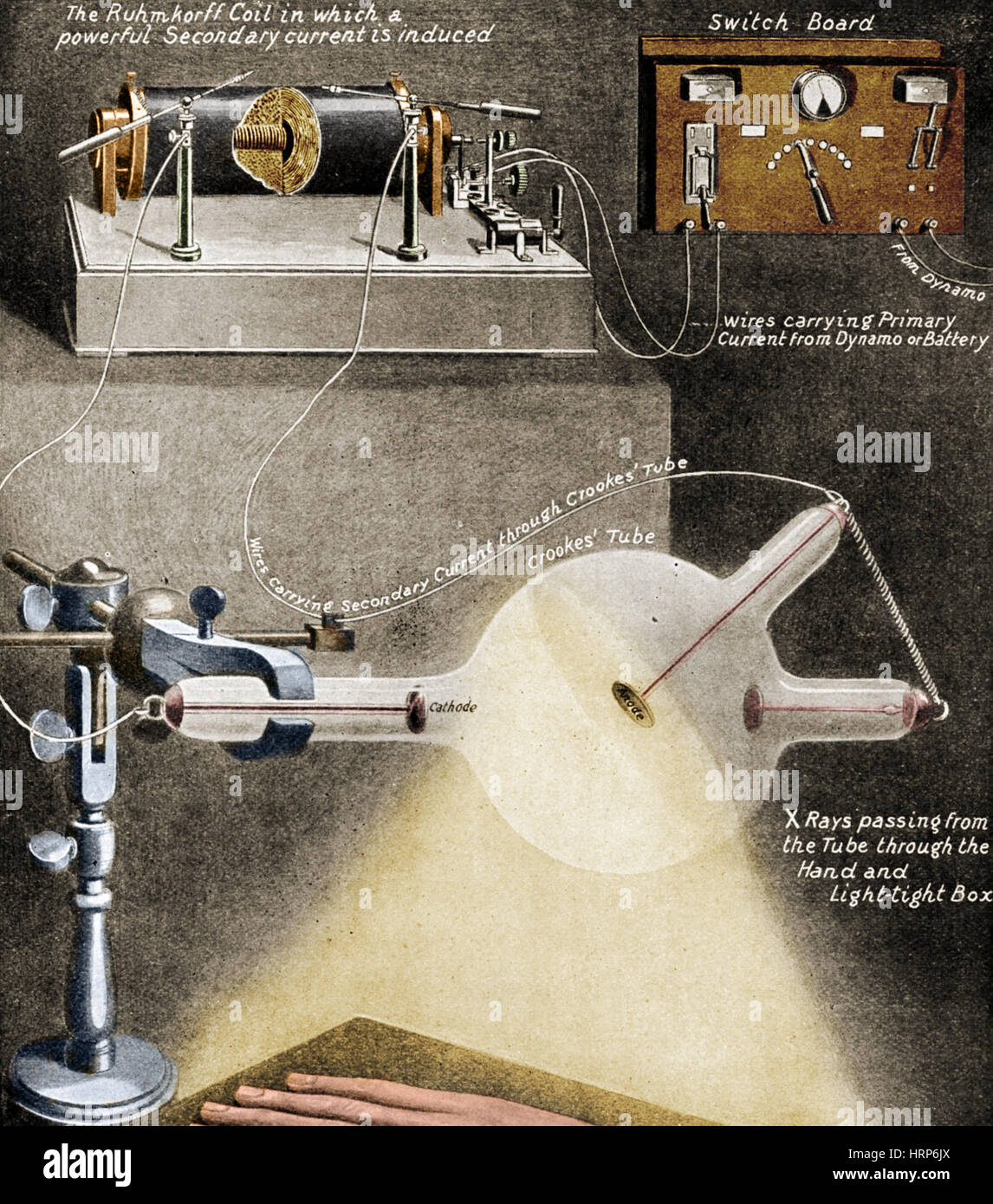 
4.1 First steps in the discovery of the proton.1 Was there an atomic model of Goldstein?.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
